EVOCA™ continues to prove its value as novel biocontrol tool to protect crops against key fungal diseases
Ghent, BELGIUM, Dec. 19, 2023 (GLOBE NEWSWIRE) -- Biotalys (Euronext – BTLS), an Agricultural Technology (AgTech) company developing protein-based biocontrol solutions for crop protection, today announced the continued strong performance of its first biofungicide candidate EVOCA™* in independent efficacy field trials conducted by highly reputed public institutes in the U.S. EVOCA performed as a true replacer for existing crop protection products to combat fungal diseases in grapevines and strawberries. The results confirm the previous findings by independent academics as published earlier on Biotalys’ website.
EVOCA aims to provide fruit and vegetable growers with a safe new crop protection product to help control the devastating fungal diseases Botrytis and powdery mildew. By offering a novel, biodegradable tool EVOCA can contribute to reducing dependency on chemical pesticides and development of pathogen resistance.
Dr. Eva Van Hende, Biotalys’ Head of Regulatory and Field Development, noted: “These independent field trials by prestigious academic institutions in the United States continue to validate the efficacy of EVOCA, our first biocontrol developed on our AGROBODY™ technology platform. We are dedicated to obtaining regulatory approval for this product and to creating an expansive pipeline of biocontrols to support the transition to safer, more sustainable agricultural practices.”
The trials were conducted in 2022 and 2023 by the University of California Davis and the University of Florida and are industry gold-standard studies that provide growers and crop advisors with detailed information on the performance of crop protection products. In these grape and strawberry trials, EVOCA was tested among many other treatments and a non-treated control plot, enabling the comprehensive comparison of its performance with conventional chemical and biological fungicide products.
The outcome of the academic studies in California is particularly relevant as California is the largest U.S. grape market. In that respect, the Californian Department of Pesticide Regulation (CDPR) has been reviewing the regulatory dossier for EVOCA submitted by Biotalys in April 2021. The company now understands that the CDPR has finalized its own in-depth review, opening a path to a swift approval at state level if the U.S. Environmental Protection Agency (EPA) approves the product at federal level.
Study highlights
- EVOCA performed as a true replacer for chemical products as well as a top performing biological option to combat Botrytis in grapes, in particular at the critical flower (or bloom) application window;
- EVOCA proved consistent protection against Botrytis in a variety of application programs for grapevines;
- EVOCA was part of the best performing biofungicide rotation scheme against Botrytis in grapes;
- In strawberry trials, rotation programs including EVOCA provided equivalent protection against powdery mildew as chemical-only rotations.
Annex: Study details
The universities publish study details on their websites. Below is a summary of results for EVOCA.
A. University of California Davis (Primary Investigator, Dr. Akif Eskalen) – grapevine trials
Botrytis fruit rot - 2023 - 389969.pdf (ucanr.edu)
Across a total of 49 different treatment programs, EVOCA (with or without adjuvant) substituted one or two chemicals in a spraying program of leading fungicide products ranked 5/5 for excellent and consistent control of botrytis bunch rot. Results confirmed EVOCA’s strong performance as a true replacer for chemical solutions, on par with the reference product Switch. EVOCA was also used in the best performing rotation program, reducing bunch rot incidence by 65% compared to a full chemical program when used in combination with an adjuvant.
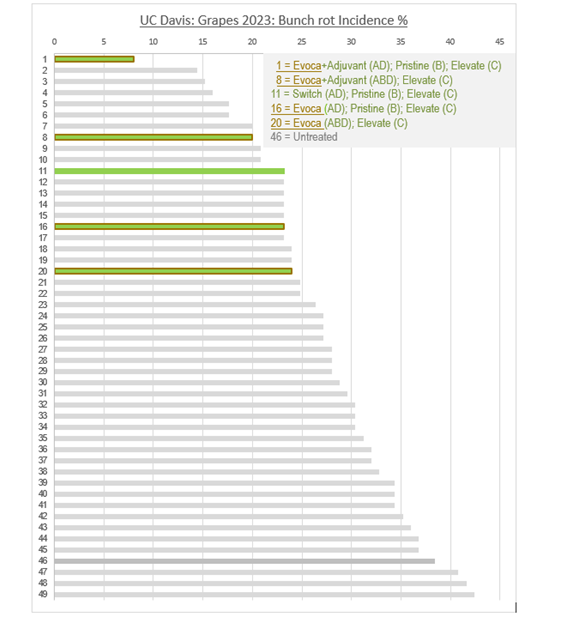
GRAPES 2023: TREATMENT SCHEME 1: Evoca+Adjuvant (AD); Pristine (B); Elevate (C)

GRAPES 2023: TREATMENT SCHEME 46: Untreated
Botrytis fruit rot - 2022 - 372339.pdf (ucanr.edu)**
Across a total of 46 different treatment programs, 6 entries contained EVOCA as a standalone treatment or integrated at flowering in a rotation program. Treatments were applied throughout the trial during the standard application windows (bloom (A), pre-closure (B), veraison (C) and pre-harvest (D)). Some of the EVOCA treatments were mixed with adjuvants.
EVOCA showed its strength in a standalone program, both with and without an adjuvant. Integration of EVOCA in a rotation program proved to be an excellent replacement for a chemical application at flowering. 5 out of the top 8 best performing application schemes contained EVOCA, earning recognition as the best performing biofungicide rotation scheme.
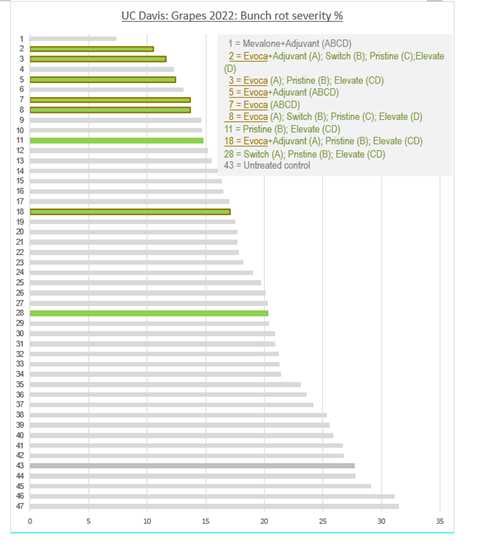
GRAPES 2022: TREATMENT SCHEME 2: Evoca+Adjuvant (A); Switch (B); Pristine (C); Elevate (D)

GRAPES 2022: TREATMENT SCHEME 43: Untreated
B. University of Florida (Primary Investigator, Dr. Natalia Peres) – strawberry trials
Botrytis fruit rot - 2022 - 2021-22-Botrytis-Fungicide-Trial.pdf (ufl.edu)***/ 2023 - Plant Disease Management Reports (plantmanagementnetwork.org)***
In 2022, EVOCA was assessed in two rotation programs at two dosages, substituting chemicals, across 17 different spraying schemes. EVOCA substitution for chemical product Captan in rotation with Switch performed as well as the reference Captan/Switch rotation, being the highest performing scheme with biofungicides.
In 2023, EVOCA was further assessed in four rotation programs, substituting chemicals, across 22 different spraying schemes. While the disease pressure was overall low and therefore does not mandate firm conclusions, the combination of EVOCA with an adjuvant was a valid substitution for Captan in rotation with reference chemical Switch or Kenja.
Powdery mildew - 2022/2023 - Plant Disease Management Reports (plantmanagementnetwork.org)***
EVOCA was assessed in two rotation programs at two dosages, substituting chemicals, across 24 different spraying schemes in both 2022 and 2023. The rotations of EVOCA with chemical products provided disease incidence levels equivalent to other chemical-only rotations.
* EVOCA™: Pending Registration. This product is not currently registered for sale or use in the United States, the European Union, or elsewhere and is not being offered for sale.
** In this study, EVOCA is identified as ASFBIOF01-02
*** In this study, EVOCA is identified as EXP14
About Biotalys
Biotalys is an Agricultural Technology (AgTech) company developing protein-based biocontrol solutions for the protection of crops and food and aiming to provide alternatives to conventional chemical pesticides for a more sustainable and safer food supply. Based on its novel AGROBODY™ technology platform, Biotalys is developing a strong and diverse pipeline of effective product candidates with a favorable safety profile that aim to address key crop pests and diseases across the whole value chain, from soil to plate. Biotalys was founded in 2013 as a spin-off from the VIB (Flanders Institute for Biotechnology) and has been listed on Euronext Brussels since July 2021. The company is based in the biotech cluster in Ghent, Belgium. More information can be found on www.biotalys.com.

For further information, please contact
Toon Musschoot, Head of IR & Communications
T: +32 (0)9 274 54 00
E: Toon.Musschoot@biotalys.com
Important Notice
Biotalys, its business, prospects and financial position remain exposed and subject to risks and uncertainties. A description of and reference to these risks and uncertainties can be found in the 2022 annual report on the consolidated annual accounts and the half-year report for 2023.
This announcement contains statements which are "forward-looking statements" or could be considered as such. These forward-looking statements can be identified by the use of forward-looking terminology, including the words ‘aim’, 'believe', 'estimate', 'anticipate', 'expect', 'intend', 'may', 'will', 'plan', 'continue', 'ongoing', 'possible', 'predict', 'plans', 'target', 'seek', 'would' or 'should', and contain statements made by the company regarding the intended results of its strategy. By their nature, forward-looking statements involve risks and uncertainties and readers are warned that none of these forward-looking statements offers any guarantee of future performance. Biotalys’ actual results may differ materially from those predicted by the forward-looking statements. Biotalys makes no undertaking whatsoever to publish updates or adjustments to these forward-looking statements, unless required to do so by law.

